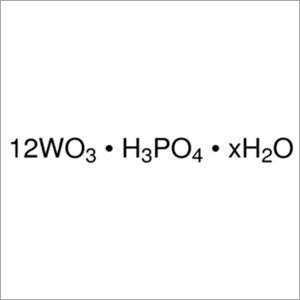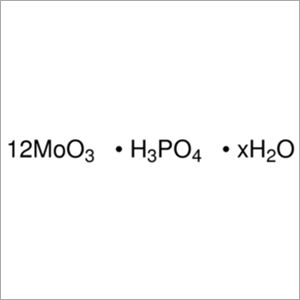Potassium Dichromate
Potassium Dichromate Specification
- Boiling point
- Decomposes before boiling
- Physical Form
- Solid
- Purity
- 99% min
- Viscosity
- Not applicable (solid)
- Grade
- Analytical Reagent (AR), Laboratory, Industrial
- Ph Level
- 1 (0.1N solution)
- Other Names
- Dipotassium dichromate
- Poisonous
- Yes, highly toxic
- Smell
- Odorless
- Density
- 2.68 Gram per cubic centimeter(g/cm3)
- HS Code
- 28415000
- Molecular Weight
- 294.18 g/mol
- Appearance
- Orange-red crystalline solid
- Structural Formula
- O7Cr2K2
- Solubility
- Soluble in water, insoluble in alcohol
- EINECS No
- 231-906-6
- Melting Point
- 398C (decomposes)
- Molecular Formula
- K2Cr2O7
- CAS No
- 7778-50-9
- Taste
- Bitter, metallic (toxic do not ingest)
- Shape
- Crystalline
- EC Index Number
- 024-004-00-7
- Decomposition Products
- Chromium oxides, potassium oxides
- UN Number
- 3288
- Stability
- Stable under ordinary conditions, but light and moisture sensitive
- Manufacturing Standard
- Conforms to AR/ACS grade
- Packaging
- HDPE jars, fibre drums, or as specified
- Safety Precautions
- Use personal protective equipment, avoid skin and eye contact, ensure good ventilation
- Uses
- Laboratory reagent, oxidizing agent, cleaning glassware, photography, chromium electroplating, dyeing, tanning
- Hazard Class
- 6.1 Toxic substances
- Storage Conditions
- Store in tightly closed containers, in a cool, dry, well-ventilated area away from combustible materials and reducing agents
About Potassium Dichromate
Available in 98% pure form, Potassium Dichromate is used to formulate pesticides and agrochemicals. This chemical has orange red color and it is non hydroscopic by nature. Its flammable characteristics are expressed when it is exposed to organic compounds. Potassium Dichromate is water soluble and its moisture content is 0.030%. Flash point and boiling point of this chemical are 50 degree C and 82 degree C respectively. Density of this substance is 7.14 g/ml at 25 degree C temperature. Its molar mass is 294.18. It easily dilutes in cold water. Long storage lie, pure composition, advanced processing technology and reasonable price are its key features.
State: Crystal
Superior Quality for Industrial & Laboratory Use
Our Potassium Dichromate is supplied in high purity (99% min) AR/ACS grade, ensuring reliable performance for both analytical and industrial applications. Each batch meets stringent international standards for quality and safety, supporting processes in laboratories, electroplating, photography, dyeing, tanning, and glassware cleaning.
Safe Packaging to Maintain Stability
Packaged in HDPE jars, fibre drums, or as specified, Potassium Dichromate remains stable under ordinary conditions when stored properly. Its light and moisture sensitivity is managed through robust packaging solutions, preventing contamination or degradation during transit and storage.
FAQs of Potassium Dichromate:
Q: How should Potassium Dichromate be stored to maintain its quality?
A: Potassium Dichromate should be kept in tightly closed containers, in a cool, dry, and well-ventilated area away from combustible materials and reducing agents to prevent deterioration and ensure stability.Q: What safety precautions are necessary when handling Potassium Dichromate?
A: When working with Potassium Dichromate, always use appropriate personal protective equipment, such as gloves and goggles, avoid contact with skin and eyes, and ensure good ventilation to minimize exposure to toxic particles.Q: Where is Potassium Dichromate commonly used?
A: Potassium Dichromate is utilized in laboratories as a reagent, for cleaning glassware, as an oxidizing agent, and in industries such as photography, dyeing, tanning, and chromium electroplating.Q: What are the benefits of choosing AR/ACS grade Potassium Dichromate?
A: AR/ACS grade Potassium Dichromate assures high purity and reliable analytical performance, making it suitable for precise laboratory experiments and demanding industrial processes.Q: What process leads to the decomposition of Potassium Dichromate and what are the byproducts?
A: Potassium Dichromate decomposes when exposed to excessive heat or moisture, producing chromium oxides and potassium oxides as decomposition byproducts.Q: When should personal protective equipment be used with Potassium Dichromate?
A: Personal protective equipment must be used at all times during handling, usage, and transportation of Potassium Dichromate due to its highly toxic nature.Q: How is Potassium Dichromate typically packaged for transport and storage?
A: It is packaged securely in HDPE jars, fibre drums, or other specified containers to protect it from light, moisture, and accidental exposure.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Fine Chemicals Category
Phosphotungstic Acid LR/AR
Minimum Order Quantity : 1 , , Kilograms
Molecular Formula : H3PW12O40
Solubility : soluble in water (200 g/100 ml)
Melting Point : 89 C
Purity : 98 %
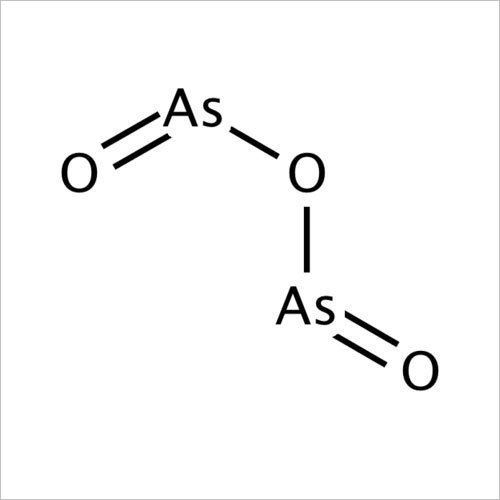
Arsenic Trioxide
Molecular Formula : As2O3
Solubility : Slightly soluble in water, soluble in alkalis and acids
Melting Point : 312 C
Purity : 99% Min.
Phosphomolybdic acid ACS AR
Minimum Order Quantity : 1 , , Kilograms
Molecular Formula : H3MO12O40P
Purity : 97 %
Nutrient Broth
Price 800.0 INR / Kilograms
Minimum Order Quantity : 100 Kilograms
Solubility : water
Purity : 98%
 |
SUVIDHINATH LABORATORIES
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |




 Send Inquiry
Send Inquiry